In the guide
Although the United Kingdom left the European Union (EU) in 2021, certain pieces of legislation (known as 'assimilated law') continue to apply until such time as they are replaced by new UK legislation, revoked or permitted to expire. This means that our guidance still contains references to legislation that originated from the EU.
In this guide, the words 'must' or 'must not' are used where there is a legal requirement to do (or not do) something. The word 'should' is used where there is established legal guidance or best practice that is likely to help you avoid breaking the law.
This guidance is for England and Wales
This guidance relates to prepacked food only. However, foods that contain meat are also subject to QUID requirements when they are non-prepacked or prepacked for direct sale.
'Prepacked food' is defined in Regulation (EU) No 1169/2011 on the provision of food information to consumers as "… food and the packaging into which it was put before being offered for sale, whether such packaging encloses the food completely or only partially, but in any event in such a way that the contents cannot be altered without opening or changing the packaging …".
The definition of prepacked does not cover food sold loose (without packaging), or food that is packaged at the request of the consumer (both referred to as non-prepacked); nor does it apply to food that is sold from the premises on which it was packed, or from a mobile stall or vehicle being operated by the packer (referred to as 'prepacked for direct sale'). For more information see 'Labelling of non-prepacked foods' and 'Labelling of prepacked-for-direct-sale foods'; it is important to understand the differences between the three types.
Quantitative ingredient declaration
Certain ingredients will need a quantitative ingredient declaration (QUID).
QUID is an indication of how much of the finished product is made up of a certain ingredient; it is always expressed as a percentage.
The QUID must either be given immediately after the ingredient appears in the name of the food or, more commonly, in brackets immediately after the ingredient appears in the ingredients list. For example, peanut butter:
- 'Peanut (95%) butter'
or - 'Ingredients: Peanut (95%), Brown Cane Sugar, Palm Oil, Sea Salt'
You may choose your preferred method.
Back to topWhen is a QUID required?
Not all ingredients will require a QUID.
Ingredients will require a QUID in the following circumstances.
The ingredient appears in the name of the food. This applies to all types of name (fancy, customary, descriptive, etc; see 'Labelling of prepacked foods: product name').
The ingredient is commonly associated with the food by consumers. If people think of a certain ingredient as being common to the product, then you must provide a QUID for it. Examples include beef mince in spaghetti Bolognese and kidney beans in chilli con carne.
The ingredient is emphasised in some way on the product. This applies to emphasis in any form, which means that the name of the ingredient (other than in the ingredients list), descriptions of the ingredient, pictures of the ingredient, etc would all trigger the need to provide a QUID. For example, a strawberry and banana smoothie that has a picture of apples on the packaging would need a QUID for the strawberry, the banana and the apple.
If you did not provide a QUID, the customer might not be able to distinguish it from products with a similar name or appearance. For example, two brands of tomato ketchup, made up of 50% tomato and 30% tomato respectively. The products may look identical and be priced similarly, so the QUID declaration allows consumers to make an easy comparison between the two products.
The above are referred to as 'QUID triggers' in this guidance.
Back to topExemptions
In certain circumstances ingredients will not need a QUID, even though they meet the above criteria.
The ingredient is used in small quantities for flavouring purposes. For example, 'Chopped tomatoes with basil and oregano'. In this example the herbs will be present in very small quantities to impart flavour and so will not need a QUID.
Even though the ingredient appears in the name of the food, the amount that is present will not govern consumer choice. A good example of this is garlic bread. Garlic appears in the name of the product and so would normally need a QUID. However, the amount of garlic present in the product will not affect a consumer's decision to purchase one product over another and, therefore, no QUID is required.
Mixtures of fruit, vegetables, mushrooms, spices or herbs where the amounts of each are likely to vary but no one ingredient dominates. These would be declared as 'fruit', 'vegetables', 'herbs', etc in the ingredients list and no QUID given.
Sweeteners, if they only appear in the name of the food because of the requirement to state 'with sweeteners'. However, if you had deliberately brought attention to the fact that you had used a certain type of sweetener - 'Now with stevia', for example - you would need to provide a QUID as normal for the stevia.
Any ingredient that already needs the quantity to be declared by other legislation. For example, cocoa solids in chocolate and fruit / sugar content in jam.
Any vitamins and minerals emphasised on the label. Any claim about vitamins and minerals triggers the need for a nutrition declaration, which will include the weight of vitamins and minerals present.
Any ingredient that has a drained weight indicated on the label.
Back to topHow to calculate the QUID
QUID is calculated at the mixing bowl stage of production.
Calculate the QUID by dividing the weight of the ingredient (X) by the combined weight of all the ingredients (Y) and multiplying by 100; the resulting number will be the percentage of the product that is made up of that ingredient.
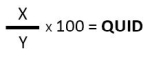
For products that lose moisture during cooking or production (for example, butter biscuits) the QUID should be the percentage of the named ingredient in the product as sold.
Back to topProducts containing meat
Products containing meat (PCMs) have their own QUID requirements, which vary depending on how the product is being sold (prepacked, non-prepacked, etc).
PCMs are foods that contain meat and any other ingredient.
Raw meat with no added ingredients (except proteolytic enzymes) and uncooked poultry with no added ingredients except additives, water, self-basting preparations or seasonings are not PCMs.
'Meat' is defined as "the skeletal muscle of mammalian or bird species recognised as fit for human consumption with naturally included or adherent tissue" (fat and connective tissue). Mechanically separated meat (MSM) and heart, tongue, etc are not meat.
A certain amount of fat and connective tissue, up to set limits (see below), is considered to be meat. Any fat and connective tissue over the set limits will not be meat and, if the permitted levels are exceeded, you will have to declare added fat and/or connective tissue in the ingredients list. Fat and connective tissue over the permitted levels, MSM, etc cannot be included when making a QUID for the amount of meat.
| Type of meat | Pork | Birds and rabbits | Beef, lamb and other species |
|---|---|---|---|
| Fat | 30% | 15% | 25% |
| Connective tissue | 25% | 10% | 25% |
For more information please refer to 'Composition of products containing meat'.
Prepacked foods
There is no absolute requirement to provide a QUID for the amount of meat in prepacked food, but it is required if one of the QUID triggers are met (see 'When is a QUID required' above).
In most cases where the product being sold is a PCM in itself (a packet of sausages, for example), a QUID will be required for the meat. This is because most foods are required to have a descriptive name (see 'Labelling of prepacked foods: product name'), which, in order to be sufficiently accurate, must specify the type of meat - for example, 'pork'. As the ingredient 'pork' has been specified in the name, the first QUID trigger is met and a declaration is required.
There will be certain foods that contain PCM ingredients where there is no requirement to specify the type of meat in the name of the food, and therefore the first QUID trigger is not met. However, if any of the other triggers are met, then a QUID for the meat ingredient will be required.
For example, consumers commonly associate lamb mince with Shepherd's Pie; therefore, the requirement to provide a QUID for the lamb is triggered, regardless of whether lamb is specified in the name of the food.
The QUID declaration must appear on the label.
Non-prepacked foods and prepacked-for-direct-sale foods
You are required to provide a QUID for the amount of meat in non-prepacked foods and prepacked-for-direct-sale foods, regardless of whether any of the usual QUID triggers are present. This is the only QUID requirement that applies to non-prepacked and prepacked-for-direct-sale food.
The information must appear either on a label accompanying the food or on a notice in close proximity to the product (a traditional notice, chalk slate, shelf-edge label, etc).
How to provide a QUID for meat
The QUID for the meat relates to the total percentage of the product made up of a named type of meat (for example, pork), at the mixing bowl stage of production.
The meat can be declared as an ingredient in the ingredients list, following all the rules for doing so (refer to 'Labelling of prepacked foods: ingredients list' for more information). For example:
- 'Shepherd's Pie:
Ingredients: Mashed Potato (Potato, Butter (Milk), Milk, Salt, White Pepper), Lamb (18%) ...'
For food that does not have an ingredients list (or optionally for those foods that do), the QUID may instead be declared with the name of the food. For example:
- 'Shepherd's Pie (18% lamb)'
Where the product contains multiple sources of the same type of meat (a product that contains both pork sausages and pork lardons, for example), you must make a single QUID declaration for that type of meat, which will be the percentage of the product from all sources of that type of meat. If we look again at the equation given in 'How to calculate the QUID' above, in this case, X is the combined weight of the meat from the sausage and the meat from the lardon, and Y is the combined weight of all ingredients (including the meat).
The meat that you include in your calculation must meet the definition of 'meat'. Any fat and connective tissue above those maximum levels permitted for each type of meat (see the above table) cannot be included in the QUID, and you must reduce the weight of the meat accordingly. To take lamb as an example, it may contain up to 25% fat and the total amount will still be considered to be meat. Therefore, 1 kg of lamb may contain 250 g of fat and still be 1 kg of lamb for QUID purposes. If your 1 kg of lamb contains 300 g of fat (30%), only the first 25% (250 g) can be included; the other 50 g must be declared as 'added fat' (or similar) and the weight of lamb will only be 950 g for QUID purposes. For example:
- 'Shepherd's Pie
Ingredients: Mashed Potato (Potato, Butter (Milk), Milk, Salt, White Pepper), Lamb (18%), Water, Onion, Carrot, Cornflour, Tomato Ketchup (Tomato, Spirit Vinegar, Sugar, Salt, Pepper Extract, Celery Extract, Pepper), Tomato Purée, Yeast Extract, Salt, Colour: Plain Caramel; Lamb Juices, Carrot Juice Concentrate, Black Pepper, Lamb Fat (Antioxidant: Rosemary Extract), Dried Onion'
If the product contains multiple types of meat, you will need to make a QUID for each type, using the rules above. For example:
- 'Chicken, bacon and leek pie (18% chicken, 4% pork)'
Exceptions
Non-prepacked and prepacked-for-direct-sale foods
The requirement to provide a QUID for the meat content of non-prepacked and prepacked-for-direct-sale foods does not apply to the following:
- raw meat to which no ingredient other than proteolytic enzymes has been added
- frozen and quick-frozen chicken with no added water
- fresh, frozen and quick-frozen poultry-cuts with no added water
- sandwiches, filled rolls and anything similar (filled bagels and croissants, for example) that are ready for consumption without further preparation (cooking, for example). However, this does not apply to burgers of any type
- pizzas and all similar topped products
- soups, broths and gravies
- any food that is an assemblage of two or more ingredients, has had no further preparation (cooking, for example) since being put together, and is sold to the consumer as an individual portion that is intended to be eaten without any further preparation (for example, sushi, appetisers and finger foods)
PCMs sold by mass caterers (restaurants, sandwich shops, etc)
'Mass caterer' means any establishment (including buildings, vehicles and stalls (whether permanent, temporary or mobile)) where, in the course of a business, food is prepared to be ready for direct consumption by consumers, whether on the premises or elsewhere. In addition to traditional restaurants and cafes, the definition includes businesses such as sandwich shops, sandwich rounds (both traditional rounds and delivery of food ordered online), food catering vehicles, and many more.
Products containing meat offered for sale to consumers by mass caterers, that are ready for consumption without any further preparation, do not need a QUID for meat content. This applies whether the PCMs are offered for sale in person or by means of distance communication, such as internet orders.
Any product that has a minimum meat content in order to meet the definition of the product must still comply with that requirement, regardless of whether the content needs to be declared (for example, a pork sausage must contain a minimum of 42% pork). For more information on the minimum meat content for certain products, please refer to 'Composition of products containing meat'.
Multiple meat content declarations
You will occasionally see products that have multiple meat content declarations. For example:
- 'Sausage Pasta Bake
Ingredients: Tomato, Cooked Penne Pasta (Durum Wheat Semolina, Water), Water, Pork (9%) Sausage (Pork (88%), Water, Fortified Wheat Flour ...)'
In this case, the food business has given a QUID for the meat (9%, because 9% of the total product consists of pork) but has additionally stated the meat content of the sausage (88%) to highlight the quality of the ingredient.
This is permitted, but not required.
Back to topFurther information
There are many requirements for prepacked food. Please see our other guides on the subject:
- 'Labelling of prepacked foods: general'
- 'Labelling of prepacked foods: ingredients list'
- 'Labelling of prepacked foods: nutrition declaration'
- 'Labelling of prepacked foods: product name'
Trading Standards
For more information on the work of Trading Standards services - and the possible consequences of not abiding by the law - please see 'Trading Standards: powers, enforcement and penalties'.
Back to topIn this update
No major changes.
Last reviewed / updated: September 2024
Back to topKey legislation
- Food Safety Act 1990
- assimilated Regulation (EU) No 1169/2011 on the provision of food information to consumers
- Food Information Regulations 2014
- Food Information (Wales) Regulations 2014
Please note
This information is intended for guidance; only the courts can give an authoritative interpretation of the law.
The guide's 'Key legislation' links go to the legislation.gov.uk website. The site usually updates the legislation to include any amendments made to it. However, this is not always the case. Information on all changes made to legislation can be found by following the above links and clicking on the 'More Resources' tab.

